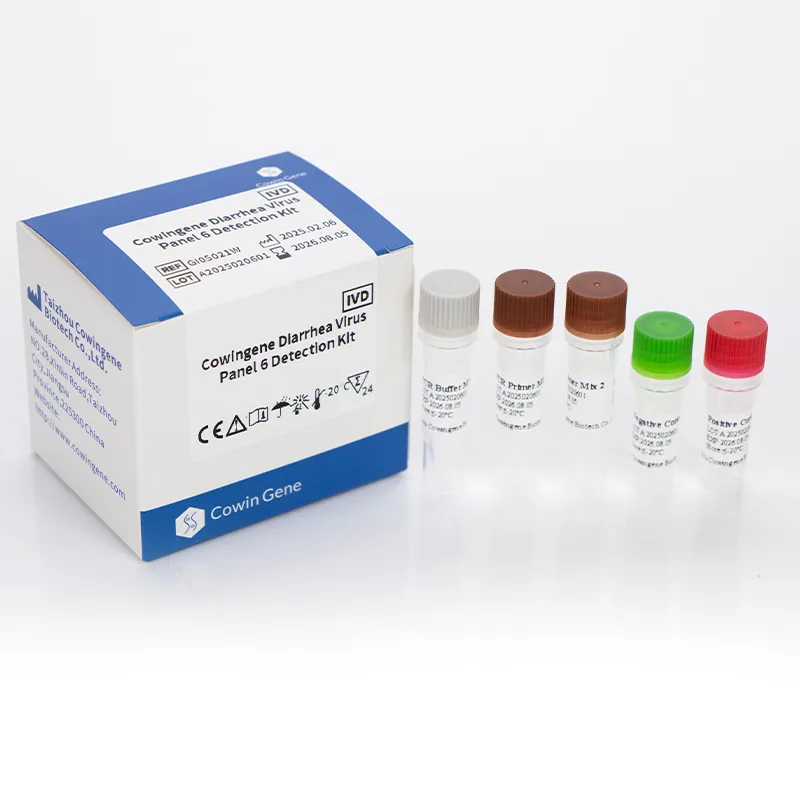
comprehensive gi panel
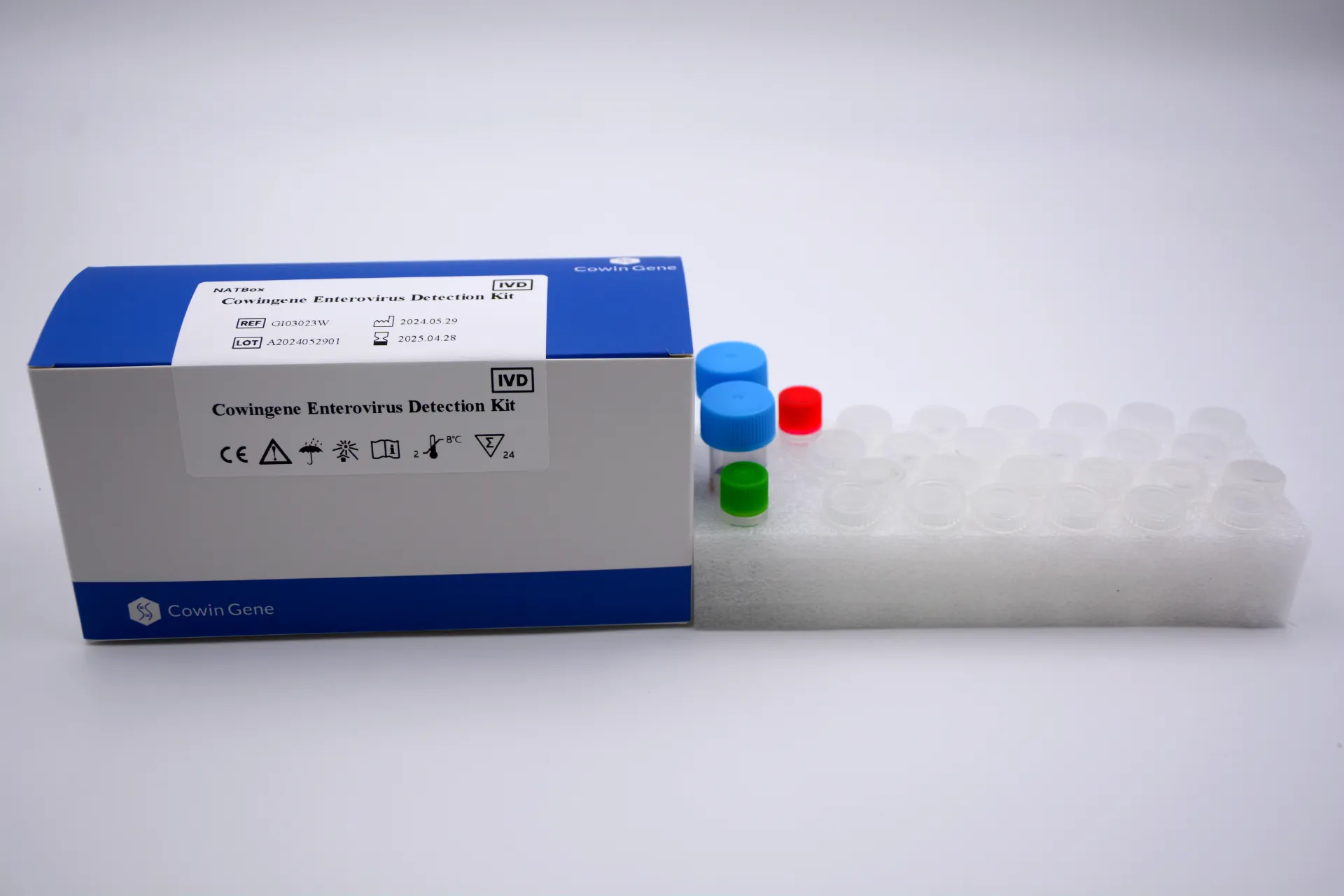
Inside the lab: a practical look at a norovirus panel that actually fits the workflow Every winter, GI outbreaks remind us how quickly a ward can grind to a halt. When teams ask me what to deploy, I point them to a well-built norovirus detection kit that doesn’t overpromise and underdeliver. In fact, multiplexing norovirus alongside other common culprits is where the time savings really happen. Product snapshot: Cowingene Diarrhea Virus Panel 6 (Lyophilized) From Jiangsu, China (NO.28, Xinlin Road, Taizhou), this lyophilized panel covers the usual suspects in acute gastroenteritis. To be honest, the freeze-dried format is what many customers say simplifies shipping and bench logistics. Parameter Details (≈ real-world use may vary) Product Cowingene Diarrhea Virus Panel 6 Detection Kit (Lyophilized), REF: GI05022U Validated specimen Feces, Vomit Analytes Norovirus GI (NoV-GI), Norovirus GII (NoV-GII), Adenovirus (AdV), Sapovirus (SV), Astrovirus (AstV), Rotavirus (RotV) Format Multiplex RT-qPCR, lyophilized reagents for ≥1 freeze–thaw resilience Throughput Scales from small batches to 96-well plates; instrument-dependent Service life Typical lyophilized kits ≈ 12–24 months when stored per IFU (check COA) How labs actually run it Materials: validated collection vessels, viral transport medium (if used), extraction kit or direct lysis buffer, RNase-free plastics, calibrated RT-qPCR system. Methods: sample pretreatment → nucleic acid extraction (or validated direct method) → master mix reconstitution → plate setup → amplification with appropriate channels → interpretation using Ct cutoffs set by the IFU. Testing standards to anchor on: CLSI MM19/13 for molecular diagnostics, ISO 15189 for medical labs, and good old internal QC with positive/negative controls each run. Industries: hospital clinical labs, public health surveillance, foodborne outbreak response, cruise lines, eldercare facilities, and central reference labs. Why multiplex, not singleplex? Because GI symptoms overlap wildly. A norovirus detection kit alone answers one question; a six-analyte panel answers the one you really care about: what’s causing this cluster today? The lyophilized format also reduces prep time—less pipetting, fewer errors. Surprisingly, that’s where a lot of time is lost in smaller labs. Real-world scenarios (condensed) District hospital outbreak triage: mixed positives for NoV-GII and RotV led to separate isolation cohorts, cutting bed closures within 48 hours. (Illustrative of common workflows.) Catering audit: negative panel across all six targets supported rapid clearance of a suspected food handler, preventing unnecessary downtime. Vendor landscape (quick take) Vendor/Format Multiplex breadth Workflow Footprint/Cost ≈ Cowingene (lyophilized RT-qPCR) 6 targets incl. NoV GI/GII Bench-friendly; uses existing qPCR Low–moderate; leverages current instruments Typical cartridge POCT 3–20 targets Simple, closed system Higher per-test; low training needs Generic liquid RT-qPCR kits 1–4 targets More pipetting; cold-chain sensitive Low per-test; higher handling time Customization and compliance Teams often ask about panel tweaks—say swapping in additional GI targets for surveillance work. It seems that custom lots, IFU localization, or RUO variants are feasible in this segment; confirm lead times and regulatory status. For clinical use, look for ISO 13485 manufacturing, CE-mark/IVD or local NMPA/FDA pathways as applicable, and lab accreditation under ISO 15189 or CAP. What users report (recurring themes) Reduced setup time vs. liquid mixes; fewer errors on busy night shifts. Clear Ct interpretation with built-in controls (always verify control behavior per run). Supply continuity matters more than flashy specs—stock up before the season. Bottom line: if your lab already runs RT-qPCR, a multiplex norovirus detection kit in lyophilized format hits a sweet spot—cost-aware, scalable, and aligned with current standards. Authoritative references CDC. Norovirus: Clinical Overview and Diagnostics. https://www.cdc.gov/norovirus/ CLSI. MM19/Related Molecular Diagnostic Standards. https://clsi.org ISO 15189: Medical laboratories — Requirements for quality and competence. https://www.iso.org WHO. Laboratory testing for norovirus. https://www.who.int FDA. Guidance on In Vitro Diagnostic Multivariate Index Assays and Multiplex Tests. https://www.fda.gov
PROFESSIONAL TEAM
Cowin Gene has a dedicated team of senior scientists, engineers and marketing experts who are committed to technological innovation and product optimization to ensure the company's leading position in the field of molecular diagnostics.
See More...LATEST NEWS